Medical Device Disinfectant: CE Mark Verification Step-by-step Guidance
Article Summary
Medical device disinfectants must be CE marked to demonstrate compliance with EU MDR 2017/745. Classification under Rule 16 determines whether a disinfectant is Class IIa or IIb, with conformity assessment, chemical, antimicrobial, and compatibility testing required to verify safety and performance. Proper technical documentation and independent laboratory testing ensure compliance, support antimicrobial claims, and protect users and devices from harm.Article Contents
What classification of medical device is my disinfectant?
Annex VIII of the EU 2017/745 Medical Device Regulation (MDR) outlines the classification rules for medical devices. Application of these classification rules should be determined by the intended use of the device. Rule 16 states that “all devices intended specifically to be used for disinfecting or sterilising medical devices are classified as class IIa”. However, under the same rule it states that if the disinfecting solutions or washer-disinfectors are specifically intended to be used for disinfecting invasive devices as the end point of processing, the device would be classified as class IIb. As a result, any liquid, spray, wipe, aerosol, or technology intended to disinfect or sterilise a medical device should apply this rule.

What conformity assessment procedures should be followed for disinfectant medical devices?
Disinfecting or sterilising medical devices are subject to conformity assessments as specified in Chapters I and III of Annex IX, including an assessment of technical documentation as specified in Section 4 of that annex (for at least one representative device for each category of devices). Alternatively, manufacturers with a class IIa device may choose to select a conformity assessment as specified in Section 10 or 18 of Annex XI, which can then be coupled with a type examination specified in Annex X for devices classified as class IIb.
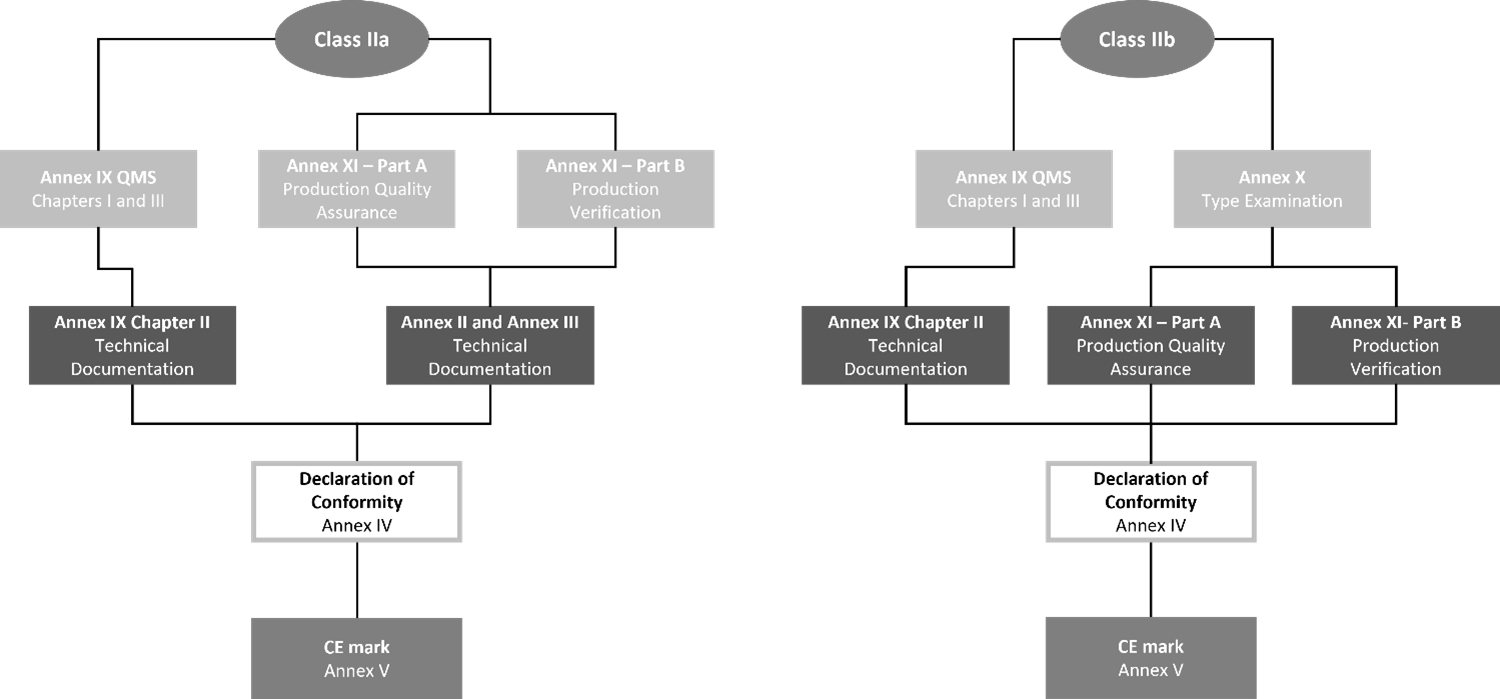
How do you validate your disinfectant medical device?
As part of the technical documentation of a disinfecting or sterilising medical device, conformity with the requirements of the MDR which are applicable to the device must be demonstrated. This includes fulfilling the requirements outlined by the relevant harmonised standards (as published in the Official Journal of the European Union). Where no harmonised standards exist or are not sufficient to demonstrate all general safety and performance requirements outlined in Annex I, common specifications may need to be adopted.
Depending on the type of product and its’ intended purpose, along with the specific claims by the manufacturer, the tests that need to be performed can vary. These tests should be carried out by a certified laboratory in accordance with the specific national or international standards which apply to the product. When performing the benefit-risk analysis, testing requirements should be determined in order to show compliance with the MDR, taking into consideration the following types of tests:
- Chemical, toxicology and efficacy testing of active ingredients and formulated disinfectants
- Antimicrobial efficacy testing
- Compatibility testing of raw materials and/or final products
Chemical, toxicology and efficacy testing of active ingredients and formulated disinfectants
For testing a disinfecting or sterilising product, BS EN ISO 10993 series of standards for biological evaluation of medical devices should be applied. Part 1 of this standard outlines the evaluation and testing requirements within a risk management process and can be used a guide to identify the biological effects of a product based on contact and duration.
When performing the biological evaluation, the manufacturer should consider the effects when the product is used as intended, as well as to eliminate or reduce risk related to user error. This identifies that a medical device which may come into contact with skin for a limited time should consider as a minimum cytotoxicity, sensitisation and irritation effects of the product.
Evidence confirming the defined physical and/or chemical specifications are met should also be included within the products’ technical documentation. Examples of this include third party confirmation of the concentration of the active ingredients within the product as well as physical, chemical, and antimicrobial stability testing.
Stability testing assesses these three aspects of a product to ensure compliance of the product for the duration of its’ shelf life, verifies the defined expiry date as well as the suitability of the products container. Aspects which can be assessed include the appearance, pH, boiling point, odour, active concentration, and efficacy.

Antimicrobial efficacy testing
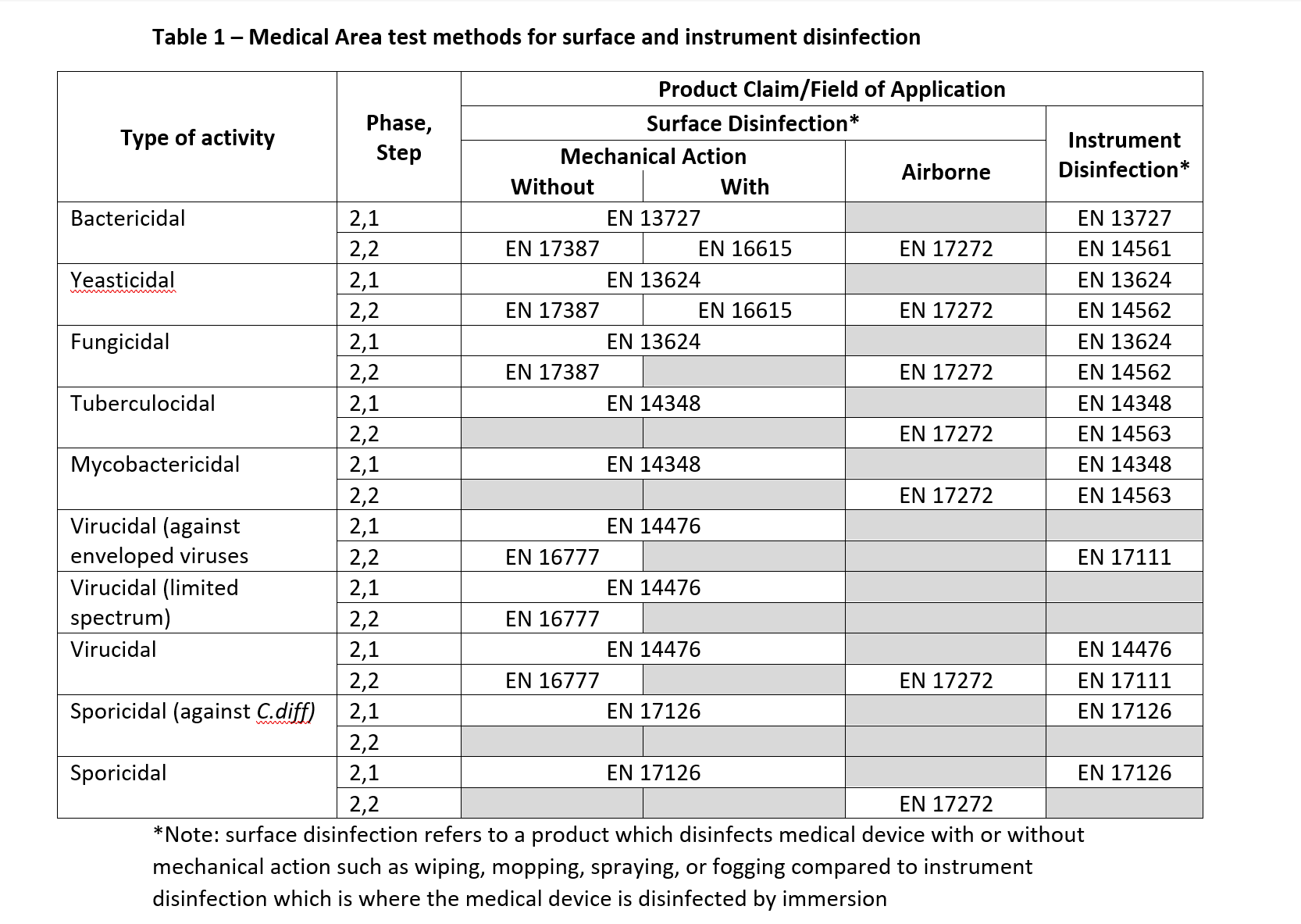
For the antimicrobial efficacy of a disinfectant, the standard BS EN 14885:2022 “Chemical disinfectants and antiseptics. Application of European Standards for chemical disinfectants and antiseptics” specifies the laboratory methods to be used to support claims appropriate to their intended use. Each standard referenced outlines the minimum requirements which should be met to pass the standard, however, is limited to a range of microbial species which have been chosen as a representative species, taking into account their relative resistance and their relevance to practical use. The manufacturer should determine an appropriate contact time, and what additional organisms should be tested to show compliance with their claims.
Independent of this standard, the manufacturer should ensure the most current version of the standard has been tested against as well as any other applicable standard which are not yet mentioned. Based on the intended purpose of the device the tests referenced in BS EN 14885:2022 (shown in Table 1) should be considered for products used in the medical area.
Compatibility testing of raw materials and/or final products
If a product is intended to disinfect or sterilise a medical device it should be designed in a way to reduce as far as possible the risk of wear, degradation, and residues. The performance requirements in the regulations must be fulfilled, including when considering the impact of processes on material properties.
Disinfecting and sterilising products have the potential to cause visual, chemical, and physical changes to exposed materials over time. This can result in the exposed medical device failing or breaking prematurely. While all manufacturers of medical devices should test disinfectant/sterilisers active ingredients to determine their own Instructions for Use in regard to cleaning, disinfecting and sterilising their product, this can not take into consideration a specific formulation of a disinfectant product or technology. This responsibility has to fall with the manufacturers of liquids, sprays, wipes, aerosols, and disinfectant technologies.
Tests on a range of medical grade materials should be performed and selected based on the mode of action of the product such as spraying, soaking (immersion), wiping or the specific technology e.g. hydrogen peroxide, UV-C light or ozone. These tests expose the raw material or a final product to the disinfectant/sterilising product to repeated cycles of use through an accelerated study to correlate to typical lifetime exposure of a product.
Another consideration is testing against BS EN 455-2 to determine glove compatibility. This will determine if the product such as a wipe (which may require latex, vinyl or nitrile gloves to be worn when being used) has a deleterious effect on the glove tensile properties.
Get It Done, With Certainty.
Contact us about your testing requirements, we aim to respond the same day.
Get resources & industry updates direct to your inbox
We’ll email you 1-2 times a week at the maximum and never share your information


