FDA 513(g) Request Explained: Benefits, Risks, and When to Use It
Article Summary
The 513(g) Request provides a binding FDA determination on device classification and regulatory requirements, but comes with fees, limited interaction, and often slow timelines. It is most valuable when regulatory status is unclear or confirmation is critical (e.g. general wellness or low-risk devices), but is not suitable for most development-stage decision-making.Article Contents
Introduction
Usually when sponsors choose to develop a medical device, a solid regulatory strategy will determine if the product is medical device under the FD&C Act and what the likely risk classification of the device will be. However, in some instances, these determinations are not that easy. In this article we will explore the current landscape of the 513(g) request and if this statutory tool is a prudent way to get regulatory and classification information.
What is the 513g Request for Information Process?
Section 513(g) of the FD&C Act provides a means for obtaining FDA’s views about the classification and the regulatory requirements that may be applicable to a medical product that a manufacturer believes may be regulated by FDA as a medical device. This provision state’s that within 60 days of receipt of a “request for information” the agency shall provide a written statement of classification, if any, of the device and the applicable requirements of the FD&C act. More specifically FDA may provide the following information:
- The general device classification regulation (if any)
- The risk classification of the device (Class I, II, or III)
- Whether a PMA, 510(k) or De Novo is or may be required
- Other requirements or guidance document applicable to the generic device type
- Determination on if the “device” is not regulated by FDA as a medical device. This could mean an un-regulated product or that the product is regulated as a drug or biologic and is under the jurisdiction of another FDA centre.

Email Device Determination
It should be noted; there exists a simple email pathway for FDA to give feedback on device classification. Sponsors may send an email to the Device Determination Division at FDA (devicedetermination@fda.hhs.gov) with prescribed information and the FDA will return comments within 7 days. However, you typically only receive clear feedback when the regulatory path and classification is cut and dry and you may be referred back to the 513g path or PreSub pathway. Also, the information provided is non-binding.
What are the Benefits of a 513g Request?
513g determinations are binding so, unlike PreSubmission and email determination requests, the classification determinations stated in the final letter can be considered the FDA’s literal interpretation of the law as it applies to your device. If your device is determined to be not regulated by FDA or to be a 510k exempt device, you can market your device without marketing authorisation and without fear of any enforcement action.
What are the Disadvantages of the 513g Request?
As stated previously, 513g determinations are binding. This can be a disadvantage if FDA returns a determination that you are not prepared to follow as a business. Unlike a PreSub, there is no opportunity to discuss the device and regulatory path with FDA. Therefore, the sole determination on regulation and classification rests on the information presented in the 513g submission. Other disadvantages include a user fee payment and delayed timelines (discussed later).
When does a 513g Request Make Sense?
Given the binding nature of a 513g request, this type of agency feedback may be warranted in the following cases:
- The manufacturer truly does not know if the device will be regulated as medical device.
- The manufacturer strongly believes the device is an unregulated general wellness device.
- The manufacturer has identified the probable class of the device and wants this binding determination in writing by FDA.
When does a 513g Request Not Make Sense
- When there is risk FDA will make a determination of which the sponsor is not comfortable.
- Determining what type of submission (510[k] or De Novo) the device will need.
- Receiving input on bench testing or clinical protocols.
513g Requests versus PreSubs
Table 1 summarises the characteristics of a 513g request and a PreSub Request.
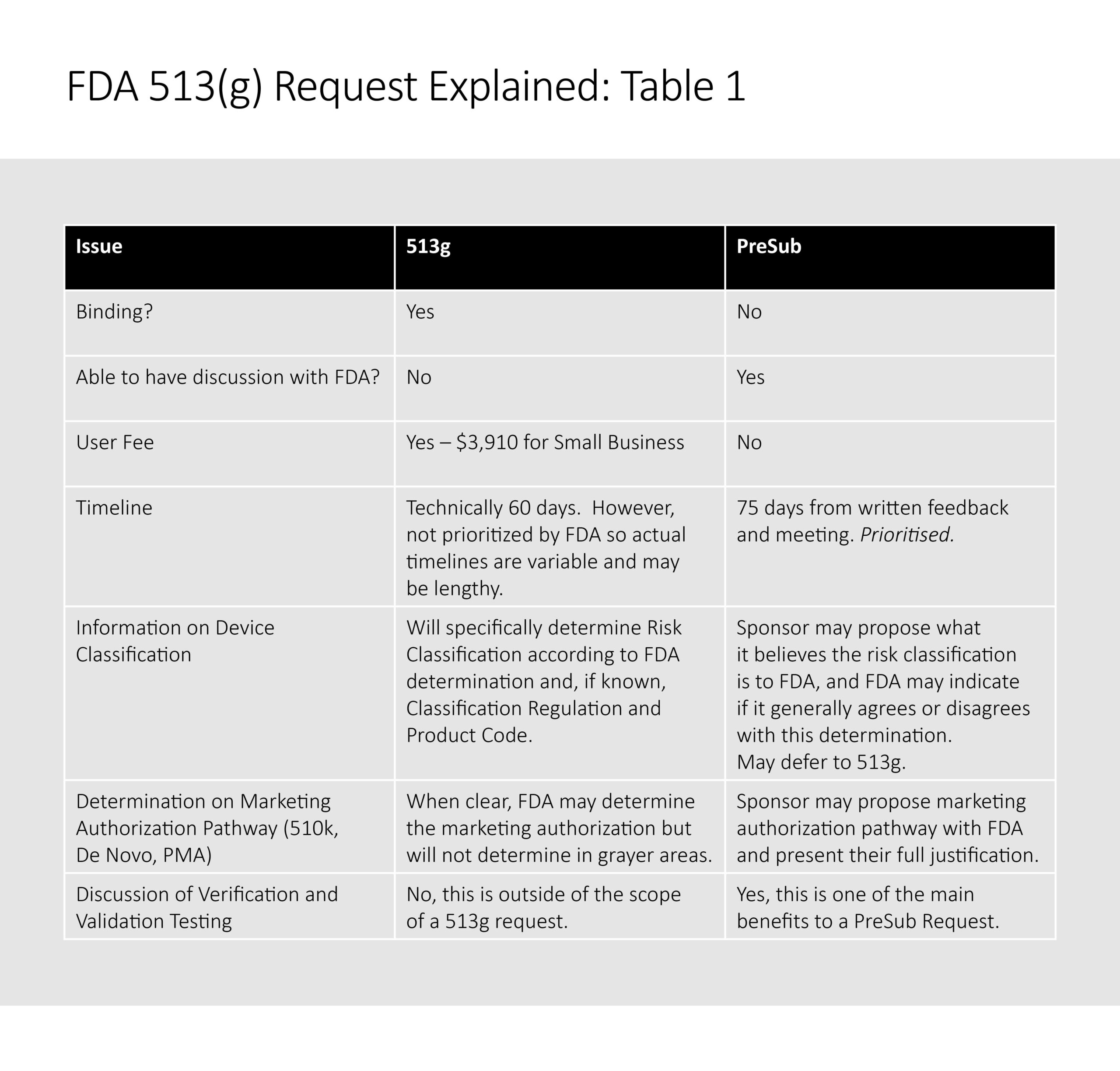
Value of the 513g Process Today
The value of submitting a 513g request and when to do it has been widely disputed and argued among medical device regulatory professionals. Recently, there appears to have been a renewed interest in sponsors using this pathway due to the increase of devices that potentially meet the FDA’s general wellness guidance. For these devices, many companies find the 513g process a good way to confirm the general wellness device status will FDA due to the binding nature of the decision to avoid potential warning letters and enforcement action down the road.
Timeframe of the FDA review of 513g Request
Though the statute expressly states that a 513g request will be revied “within 60 days” the reality could not be farther from the truth. Though the sponsor is required to pay a user fee, it is not held accountable by Congress to hit these timelines as 513g requests are no longer tracked in the annual MDUFA performance reports as of FY 2025. Therefore, FDA does not prioritise 513g requests which can leave sponsors frustrated. Available performance data from the last available MDUFA report that track 513g requests show poor review performance (Table 2).
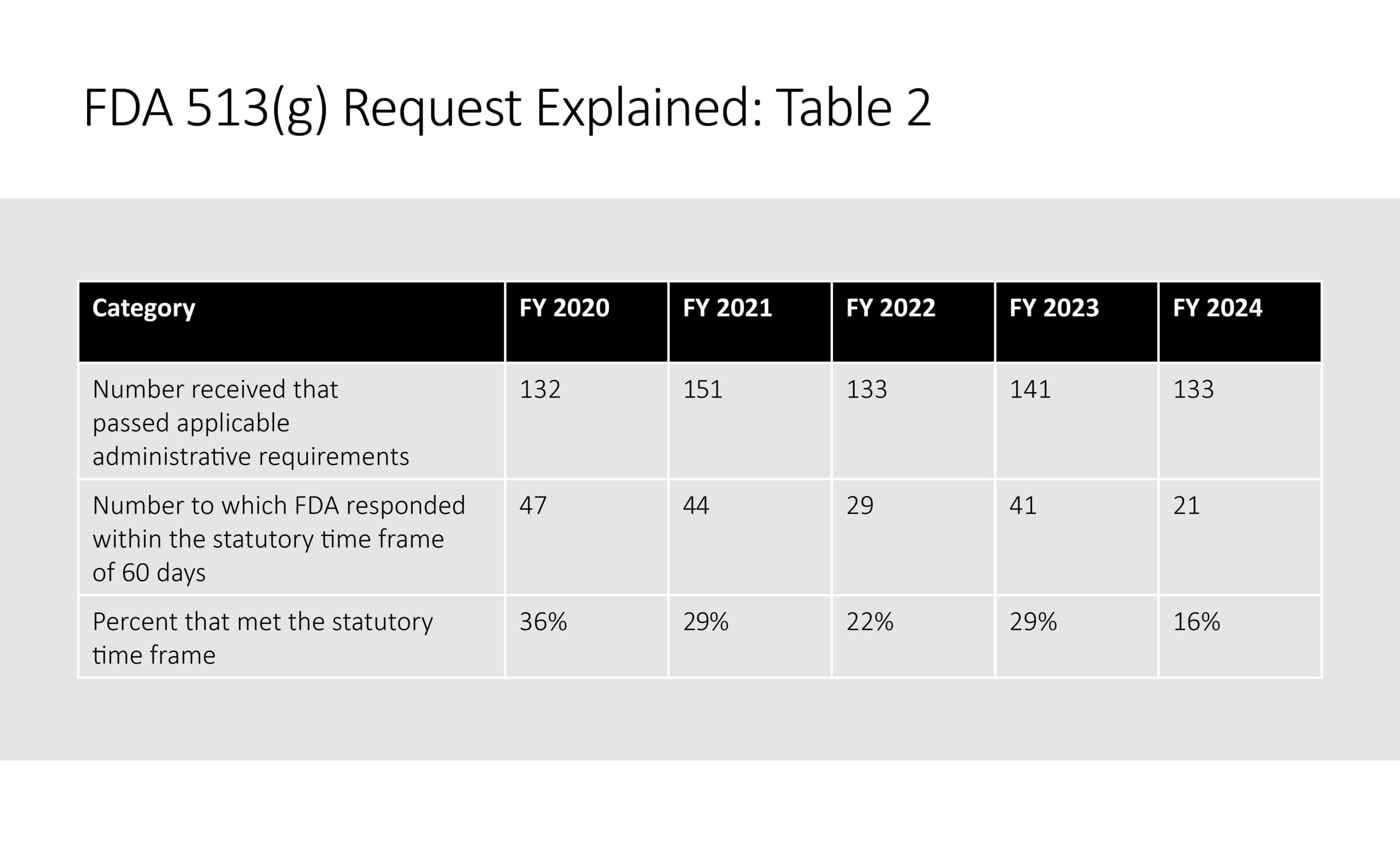
As of 2024, only 16% of 513g requests were reviewed within the prescribed 60-day time period which emphasises the lack of priority that FDA gives 513g request.
Real World Examples
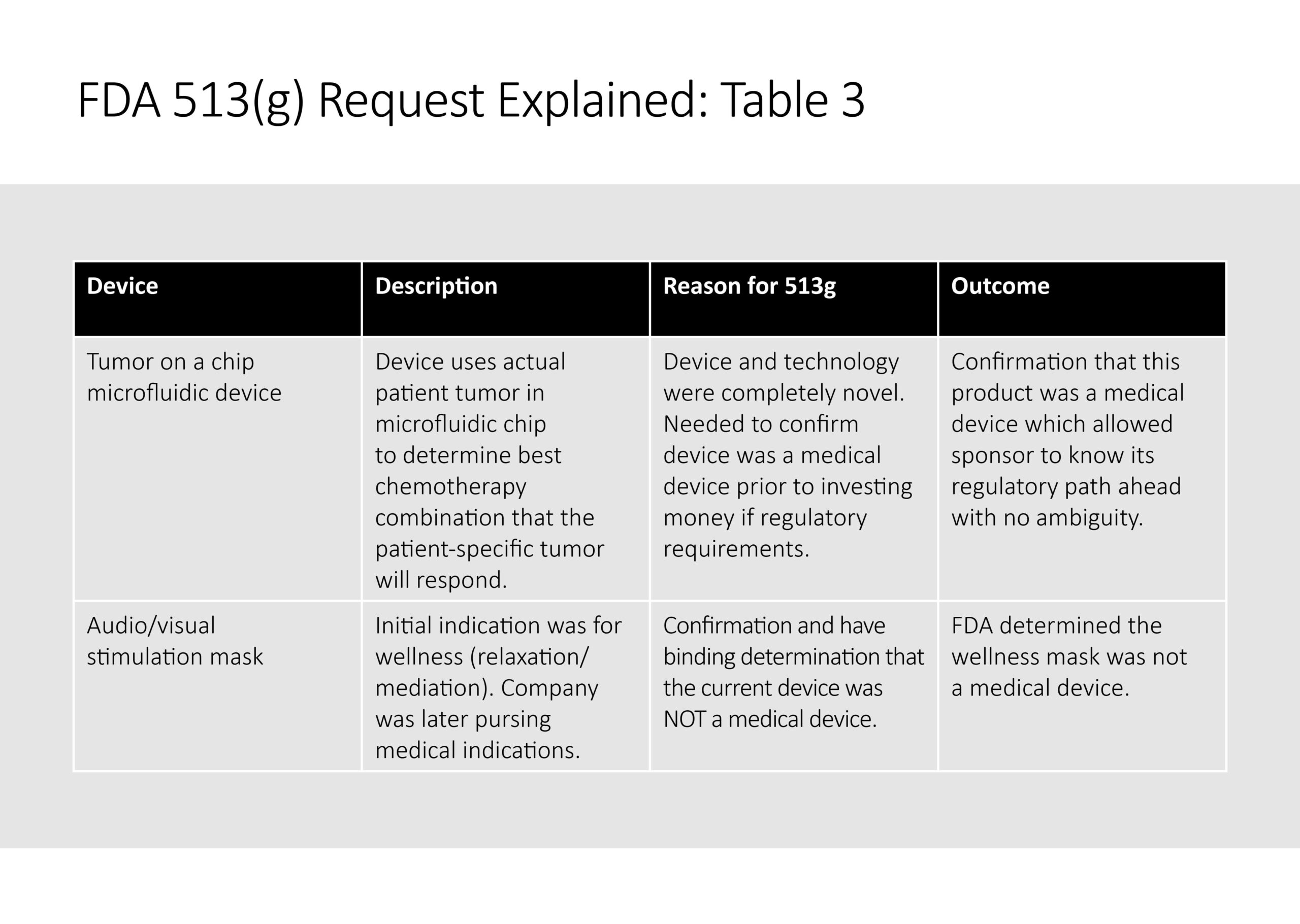
Table 3 illustrates two instances of where the 513 g was used and outcomes.
Endnote
Due to lack of prioritisation by FDA, user fee payments, and the inability to interactively discuss regulatory and classification rationale with FDA, the 513g request should be strategically and carefully considered by a sponsor and is not an ideal tool for the majority of devices. However, there are a handful of situations where a binding decision is useful to a medical device sponsor, including general wellness devices and Class I devices. The decision thus becomes a business decision.
Disclaimer. The views and opinions expressed in this article are solely those of the author and do not necessarily reflect the official policy or position of Test Labs Limited. The content provided is for informational purposes only and is not intended to constitute legal or professional advice. Test Labs assumes no responsibility for any errors or omissions in the content of this article, nor for any actions taken in reliance thereon.
Get It Done, With Certainty.
Contact us about your testing requirements, we aim to respond the same day.
Get resources & industry updates direct to your inbox
We’ll email you 1-2 times a week at the maximum and never share your information
