The Carbon Shift: Leveraging Technology-Grade Grown Diamond (TGGD) in High-Stakes Medical Hardware
Article Summary
Technology-grade grown diamonds (TGGD) are emerging as a high-performance material in MedTech. While the benefits are significant, challenges around manufacturability, cost, and regulatory validation must be addressed before widespread adoption.Article Contents
Introduction
For decades, the medical technology industry has relied on a reliable but limited palette of materials: titanium for its biocompatibility, stainless steel for its rigidity, and various polymers for their flexibility. However, as we enter the era of ultra-miniaturised electronics and precision bio-interfaces, these legacy materials are reaching their physical “ceiling”.
In the testing labs and R&D centres of leading MedTech firms, a new material is emerging: lab-grown diamonds (LGD) Technology-grade grown diamond (TGGD), synthesised via Chemical Vapor Deposition (CVD) and High Pressure / High Temperature (HPHT), is transitioning from a high-cost niche material to an emerging material being explored for next-generation medical hardware applications. However, this transition is not without significant engineering and regulatory hurdles.

Thermal Management: The Medtronic DiamondTemp Case Study
One of the most immediate applications of technology-grade grown diamond (TGGD) in clinical practice is solving “thermal lag” during high-power procedures. In cardiac ablation – specifically for treating atrial fibrillation (AFib) – the goal is to create precise lesions in heart tissue using radiofrequency (RF) energy.
Traditional metal-tipped catheters often struggle with real-time temperature accuracy because blood flow cools the tip, creating a discrepancy between the recorded temperature and the actual tissue temperature. This can lead to “char” formation or “steam pops” which are significant safety risks.
Medtronic’s DiamondTemp™ Ablation System represents a paradigm shift here. By utilising an industrial-grade diamond tip, the device leverages diamond’s exceptional thermal conductivity (which, at ~2,000 W/mK, is over five times that of copper). This allows for:
- Rapid Heat Dissipation: Ensuring the electrode remains cool while the tissue is heated.
- Real-time Feedback: The diamond tip acts as a thermal bridge, providing nearly instantaneous temperature readings.
Technical Critique: While thermally superior, diamond material can be brittle. Unlike platinum-iridium tips that can withstand mechanical deformation, TGGD components are susceptible to fracture under extreme mechanical stress. Engineers must account for these failure modes through specialised structural support and housing.
Precision Surgery: Beyond the Steel Scalpel
In ophthalmology and neurosurgery, the geometry of an incision determines the speed of the healing process. While stainless steel is the industry standard, its crystalline structure prevents it from being sharpened to an atomic level without losing its edge after a single use.
Companies like Corza Ophthalmology and Mastel Precision have integrated TGGD into their surgical lines. These diamond-bladed knives offer an edge radius of approximately 5 nanometres – roughly 100 times sharper than a premium steel blade.
Challenges in Scalability: Despite the clinical benefits – lower penetration force and cleaner incisions – manufacturing scalability remains a barrier. Producing consistent, defect-free diamond edges at a high volume requires specialised laser ablation and polishing processes that are currently more time-intensive than traditional steel stamping or grinding.
The Neural Frontier: Boron-Doped Diamond (BDD)
As we move toward closed-loop neural interfaces, the electrochemical stability of the material becomes paramount. Boron-Doped Diamond (BDD) transforms diamond from an insulator into a high-performance semiconductor.
BDD electrodes offer a unique “electrochemical potential window”, allowing them to detect biological markers (like dopamine and serotonin) without causing electrolysis in surrounding tissue.
Regulatory and Longitudinal Hurdles: While research into ultra-nanocrystalline diamond (UNCD) coatings suggests they could last 50+ years, there is currently a lack of long-term in vivo human data. To move from lab to clinical implant, manufacturers must navigate rigorous ISO 10993 biocompatibility standards and provide multi-year longitudinal studies to prove the hermeticity and stability of diamond coatings within the corrosive environment of the human body.
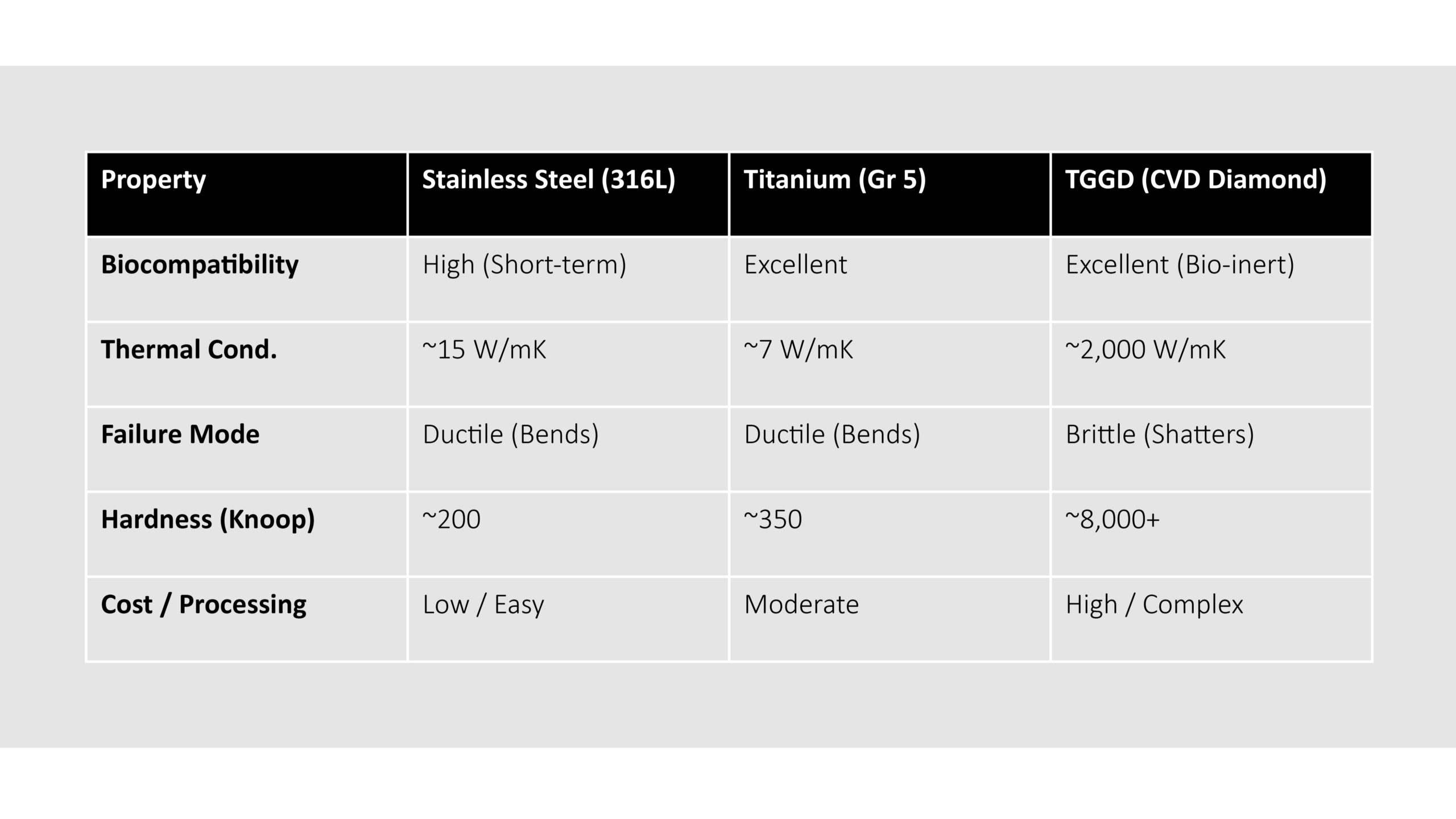
Material Comparison: Steel vs. Titanium vs. TGGD/Diamond
Implementation Challenges for MedTech Engineers
The transition to technology-grade grown diamond (TGGD) requires a rethink of manufacturing workflows:
- Specialised Fabrication: Diamond cannot be machined using traditional CNC methods. It requires laser ablation or specialised grinding, often performed by boutique houses.
- Regulatory Path: Proving that a synthetic diamond component meets the stringent safety requirements for permanent implants requires extensive testing for Foreign Body Response (FBR).
- Cost vs. Performance: The “total cost of ownership” must be justified. While diamond reduces procedure times and improves outcomes, the initial capital expenditure remains a hurdle for mid-market medical devices.
Endnote: The New Carbon Standard
The integration of technology-grade grown diamond (TGGD) is increasingly becoming a question of how and when the industry can solve the remaining material science and regulatory puzzles. While the “Carbon Revolution” promises devices that are biologically invisible, the road to the 2030s requires a balanced approach that respects both the unmatched performance of diamond and the rigid safety requirements of the MedTech world.
Disclaimer. The views and opinions expressed in this article are solely those of the author and do not necessarily reflect the official policy or position of Test Labs Limited. The content provided is for informational purposes only and is not intended to constitute legal or professional advice. Test Labs assumes no responsibility for any errors or omissions in the content of this article, nor for any actions taken in reliance thereon.
Get It Done, With Certainty.
Contact us about your testing requirements, we aim to respond the same day.
Get resources & industry updates direct to your inbox
We’ll email you 1-2 times a week at the maximum and never share your information
