Dental instrument IFU validation
Test Labs was contracted out to test and evaluate the instructions for use (IFU) of a re-processable dental medical device (MD) as required by ISO 17664-1:2021 “Processing of health care products — Information to be provided by the medical device manufacturer for the processing of medical devices”. Learn more about the requirements here. This case study describes the process that Test Labs team followed to deliver IFU validation results in under 4 weeks from receiving the test samples into the laboratory.
The validation of IFU is a requirement of the MDR EU 2017/745, where the processes described in the IFU are tested to ensure they are suitable for the MD. Even when these processes are suitable in the short term, repeated reprocessing may affect the intended use of the MD: this was a main concern for the customer.
The aim of our team was to provide definitive and objective evidence of the IFU’s efficacy with a focus on the sterilisation and cleaning instructions as per the following standards:
- ISO 17665-1:2006 “Sterilization of health care products”
- ISO 15883-5:2021 “Performance requirements and test method criteria for demonstrating cleaning efficacy”
Additionally, the effect of multiple reprocesses was also assessed on the dental MD to provide a definitive answer as to whether repeated reprocessing would have an adverse effect on the MD’s intended use. More information about the MDR requirements here.

Sterilisation of health care products using moist heat: BS EN ISO 17665-1:2006
The most common methods of medical device sterilisation involve thermal, chemical or radiation treatment. Thermal sterilisation, referring to either moist heat via an autoclave or dry heat via an oven, is the most accessible sterilisation process due to the availability of thermal disinfection equipment in medical settings.
The IFU for the MD we tested outlined a thermal sterilisation method using moist heat: specifically, to autoclave the MD at 134°C for 3 mins. Our role at Test Labs was to validate the sterilisation process of the IFU using our in-house method based on BS EN ISO 17665-1:2006.
We did this by proving a minimum sterility assurance level (SAL) could be achieved by sterilisation via any autoclave capable of maintaining 134°C for at least 1 minute. The validation process was based on the following method.
The overkill method (partial cycle approach)
The overkill method is designed to achieve a reduction in excess of the SAL specified by the IFU. This method has two approaches: full cycle and partial cycle. For our testing we used a partial cycle approach, which involves a reduced level of treatment delivered to a defined organism.
In EN ISO 17665-1:2006, an overkill process is described as achieving, at minimum, a 12-log reduction (Fbio12) of biological indicators (BIs) that have a D value of 1 minute at 121.1 °C. Therefore, use of BIs with D values greater than 1 minute may invalidate the description of the overkill method when microbiological resistance is considered.
When selecting an appropriate BI that is appropriately robust for use in validation of a F0 value, the following equation can be used:
Fbio value = LOG(BI population) × D121.1 °C value
e.g., Fbio value = LOG(1.0 × 106) × 1.0 minute = 6 minutes
An Fbio value of 6 minutes is required for successful demonstration of process validation using the partial cycle approach, where a Log 12 reduction is to be achieved.
Choice of microorganism/biological indicator
The biological indicator used in testing of the IFU for the dental MD was of Geobacillus stearothermophilus ATCC 7953. However the IFU parameters outlined a cycle of 134°C for 3 mins, therefore the following equation was used to bridge the gap:
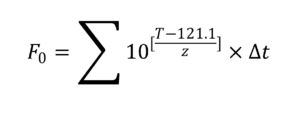
where:
F0 is the F value when z is 10 °C;
t is the chosen time interval, in minutes;
T is the temperature in the load, in degree Celsius
To calculate the equivalent time relative to a different temperature, resolve the above formula for t.
E.g.
The equivalent to 121.1 °C for 6 minutes for a 134 °C temperature would be:
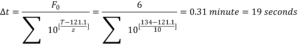
So, to achieve a log 12 reduction for a full cycle at 134 °C of 4 or 3 minutes, 1 minute deactivation of 106 spores would suffice to pass the standard.
As such, the dental MD were inoculated with 106 spores and sterilised at 134 °C for 1 minute, failure to recover any Geobacillus stearothermophilus spores was enough to demonstrate the IFU’s compliance with BS EN ISO 17665-1:2006.

Performance requirements and test method criteria for demonstrating cleaning efficacy: BS EN ISO 15883-5:2021
A washer-disinfector (WD) is by far the most common and efficient piece of equipment for routinely cleaning general equipment such as glassware. However, when a WD is used to clean more specialised equipment such as re-usable medical devices and other items used in medical, dental, pharmaceutical, and veterinary practice the cleaning efficacy of the WD and their accessories must be demonstrated.
However, not all MD are suitable to be cleaned via an automated WD and describe an alternative cleaning method within their IFU, which must also be validated. For this specific case the cleaning process of the IFU utilised manual cleaning rather than a WD.
Test Labs has developed an in-house method based on BS EN ISO 15883-5:2021 for validating IFU cleaning processes which can be modified to accommodate IFUs that do not use WDs. BS EN ISO 15883-5:2021 outlines a variety of methods and considerations specific to the type of medical device being tested. The basic approach outlined in the standard would be to soil the medical device being tested and clean it according to the specifications outlined in the IFU being validated. Following which, the presence/absence of specific analytes on the medical device would verify cleaning efficacy in a quantitative form.
Method
The MD were soiled using Edinburgh soil as described in HTM-01-01 as a worst-case scenario, then the IFU manual cleaning process was performed, in this case using a mild detergent and a soft non-abrasive brush. After the manual cleaning the efficacy of the process was confirmed in two ways:
- Visual assessment:
Visual assessment of the dental MD was performed according to our in-house test method utilising a colour assessment cabinet to ensure an even and neutral distribution of light for best possible representation of the medical device. This is also described within the ISO 21530:2004 “Dentistry — Materials used for dental equipment surfaces — Determination of resistance to chemical disinfectants”. Once this parameter was successfully met the next phase was commenced.
- Modified OPA method
The modified ortho-phthalic dialdehyde (OPA) test method is a quantitative method for the determination of primary (1°) amino groups, which are present in protein at the amino terminus of the peptide (α-) and on the amino acid lysine (ε-). OPA, in the presence of N,N-dimethyl-2-mercaptoethylammonium chloride and a primary amino group, forms stable fluorescent alkylthio-2-alkylisoindoles which is detected spectrophotometrically at 340 nm. Primary amines are ubiquitous in nature and can be present in process chemicals used in processing, therefore, negative controls are included to identify possible interfering substances. This method would quantify the residual protein left on the MD.
Additionally, a second analyte was also assessed as described in BS EN ISO 15883-5. This was ATP.
The parameters to be met were as follows:
Protein residue
- Alert level ≥ 3 μg/cm2
- Action level ≥ 6,4 μg/cm2
Adenosine triphosphate (ATP)
- Alert level ≥ 10 femtomoles (fmol) of ATP/cm2
- Action level ≥ 22 femtomoles (fmol) of ATP/cm2
The maximum acceptable level of protein on a cleaned product needed to be lower than the action level.
Performance requirements of multiple reprocessing cycles
Once all the testing was concluded and validated, for both the cleaning and sterilisation processes, the MD were then tested on multiple cycles to assess that repeated reprocess would not affect the intended use.
This was achieved by reprocessing the dental MD 50 times for cleaning and 50 times for sterilisation. After the 50 reprocessing cycles the intended use was assessed and compared to a brand new unprocessed MD. If it could be demonstrated that there was no difference in performance between the processed and unprocessed MD this would indicate the suitability of multiple reprocessing cycles and therefore full validation of the customer IFU.
Following successful validation of the IFU with BS EN ISO 17665-1:2006 and BS EN ISO 15883-5:2021 and with multiple cycles (50 in total) the IFU for the dental MD can be considered to be in compliance with ISO 17664-1:2021, fulfilling one of the requirements of the MDR.
Terms and definitions
Biological indicator
Test system containing viable microorganisms providing a defined resistance to a specified sterilisation process.
D value
Time or dose required to achieve inactivation of 90% of a population of the test microorganism under stated conditions.
Exposure time
Period for which the process parameters are maintained within their specified tolerances.
F0 value
Microbiological lethality of a sterilisation process expressed in terms of the equivalent time, in minutes, at a temperature of 121.1 °C. With reference to a microorganism with a Z value of 10 °C.
Note: An F0 of 10 minutes at 121.1 °C would be equivalent to 1 minute at 131.1 °C.
Moist heat
Thermal energy in the presence of moisture provided as steam or liquid water for the purpose of achieving microbial lethality.
Sterility assurance level (SAL)
Probability of a single viable microorganism occurring on an item after sterilisation.
Note: SAL takes a quantitative value e.g., a SAL of 10-6 offers greater assurance of sterility than a SAL of 10-3.
Z value
Temperature change required to affect a tenfold change in D value. When referring to F0 then a Z value equal to 10 °C is used.
Action level
Value from monitoring that necessitates immediate intervention.
Alert level
Value from monitoring providing early warning of deviation from specified conditions.
Analyte
Chemical substance that is the subject of chemical analysis.
Clean
Visually free of soil and below specified levels of analytes.
Soil
Natural or artificial contamination on a device or surface following its use or simulated use.
Test soil
Formulation designed for use as a substitute for a contaminant or debris found on a device after use.


