Using Device Registries to Generate Clinical Evidence for EU MDR Certification: Specific Devices
Article Summary
Medical device registries are becoming an increasingly important source of real-world clinical evidence under EU MDR, though data quality, completeness, and consistency vary widely across therapeutic areas. While orthopaedic registries offer mature, high-quality datasets, many cardiovascular, spinal, and emerging registries still face limitations that restrict their use as standalone evidence for regulatory compliance.Article Contents
Orthopaedic Joint Replacement Implants (JRI e.g. Hip, Knee, Shoulder)
Orthopaedic JRI registries (e.g., UK NJR, RIPO, NZOA Joint Registry, AOANJRR) are amongst the most advanced globally, with decades of longitudinal data, high coverage, and systematic reporting. These registries also report on other joints such as ankles and elbows. The UK NJR (covering England, Wales and Northern Ireland) has around 4.5 million procedure records submitted, with information on hip, knee, ankle, elbow and shoulder joint replacement surgery. The NJR is mandatory for all NHS trusts, foundation trusts, and independent sector hospitals in England and Wales, as well as in Northern Ireland. The NJR Minimum Data Set (MDSv8) collects patient details (with consent), hospital/surgeon information, and specific implant details. Tables 1 and 2 below summarise some data from of the JRI registries of primary procedures of the joints included in the registries. As noted in Part 1, there is some variability in how the data is reported and stratified in these registries. The registries also report on other data in addition to survival / revision rates such as clinical indications (e.g. osteoarthritis, trauma, rheumatoid arthritis), reasons for revision, patient demographic data, device details (e.g. variants, combinations, fixation, materials, sizing), hospital / centre data and PROMs; this can allow outlier detection and action can be taken if needed on specific devices, combinations, centres or users.
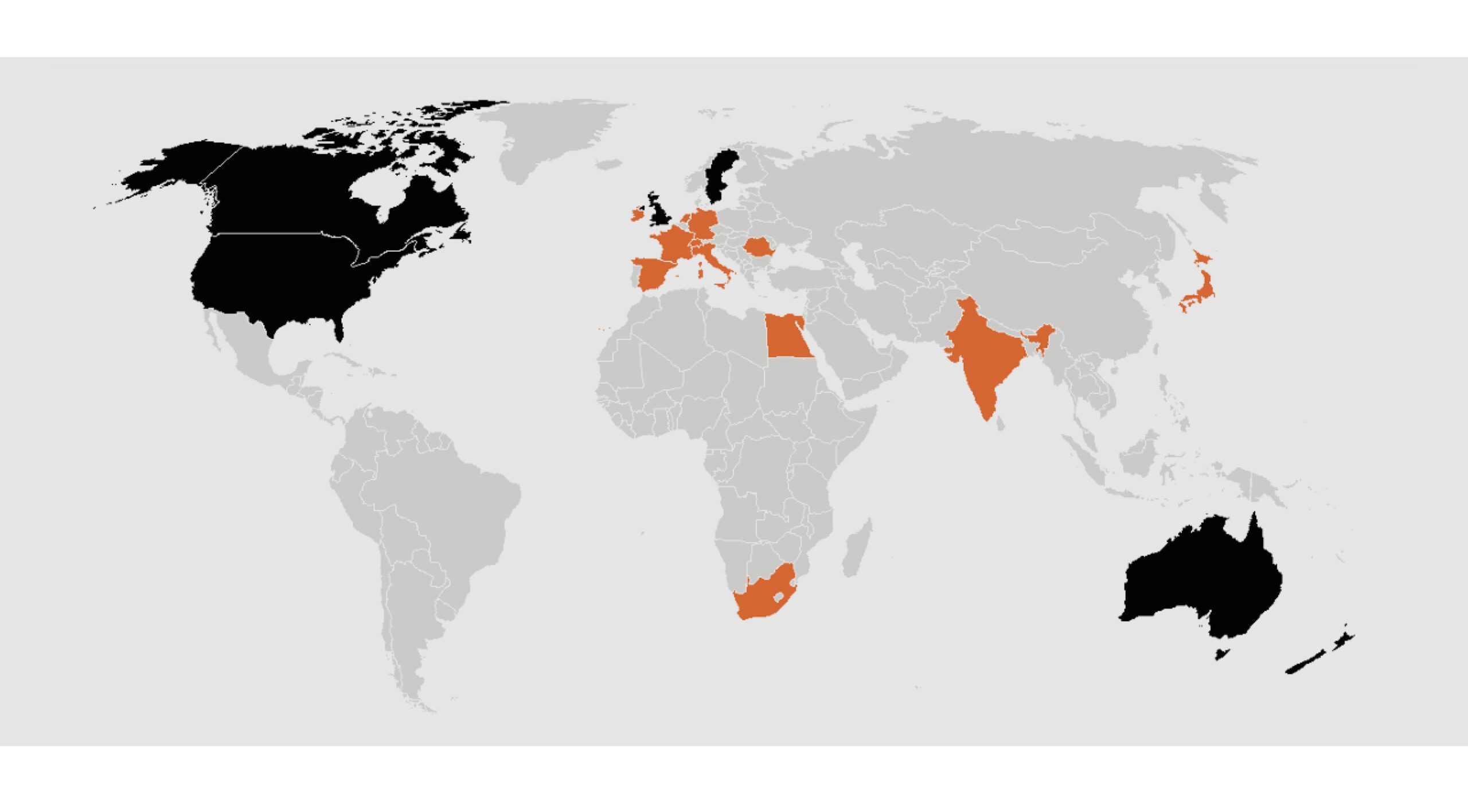
Figure 1 – countries with arthroplasty registries (Lübbeke, 2025; countries in black publish public reports with data summarised in Table 1 )
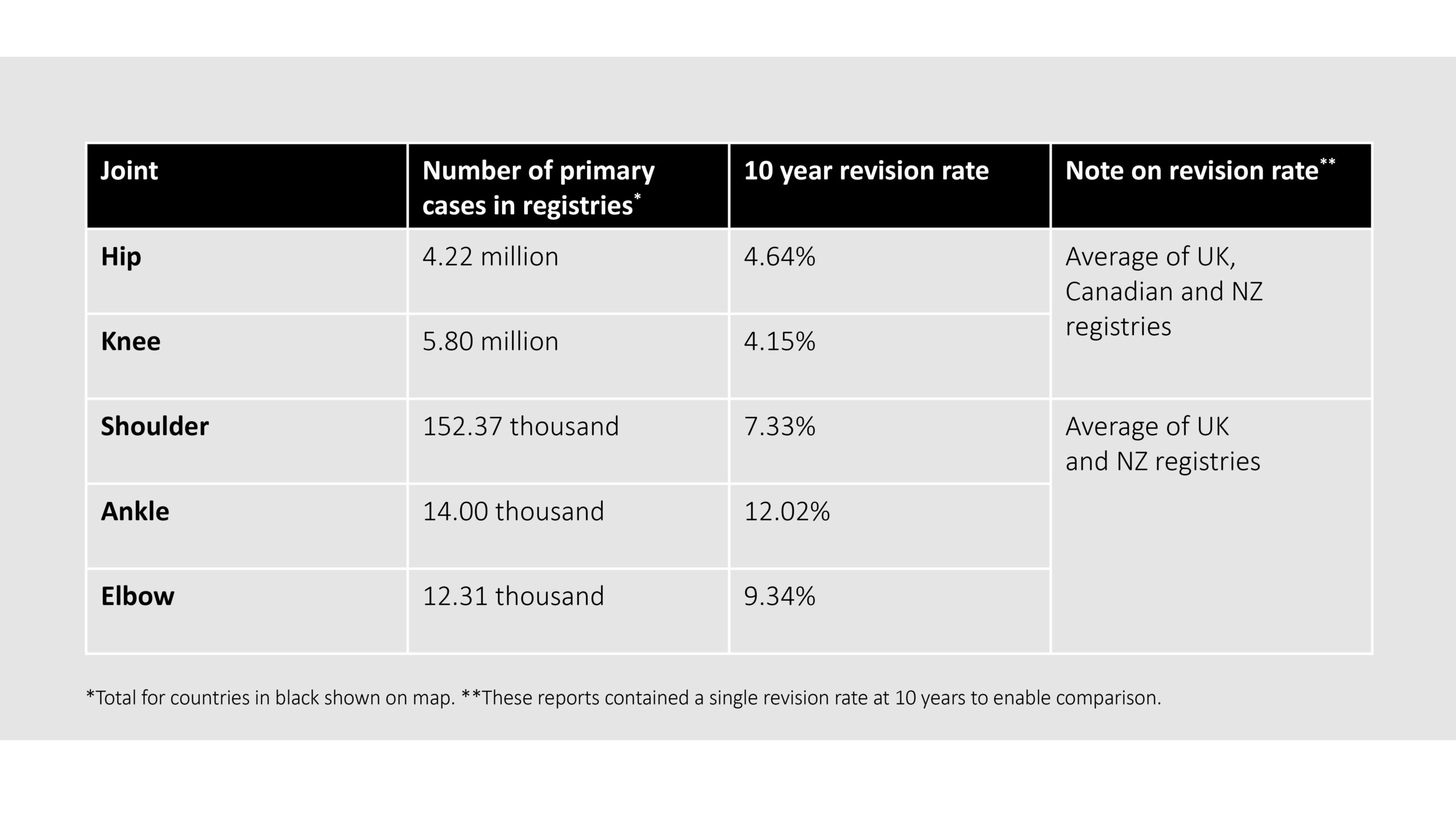
Table 1 – summary of selected primary hip and knee arthroplasty revision data from the public reports of various national registries
Hip and knee joint replacement implants are clinically well established, with mature implant designs demonstrating consistently high success rates, reflected in primary revision rates of less than 1% per year. In contrast, shoulder, ankle, and elbow arthroplasty procedures are performed less frequently and are associated with higher revision rates. This is attributable to several factors, including greater anatomical complexity and distinct biological and mechanical challenges unique to these joints.
Other arthroplasty national, regional, hospital or provider registries have been established in Ireland, The Netherlands, France, Spain, Italy, Germany, Switzerland, Romania, India, Egypt, South Africa and Japan (Lübbeke, 2025); highlighted in orange in the map in Figure 1. These registries cover hip, knee, shoulder, ankle, elbow, spine and hand procedures. Hospital coverage and completeness (of primary hip/knee procedures) is generally high (many >90%). Many of these registries also collect data on revisions, re-operations, PROMs and outliers and some share data for research with external parties.

Cardiovascular Devices (Stents, Valves, Structural Heart Devices)
A systematic review of European cardiovascular registries showed median reporting quality of only 33% of key items. None reported patient‑level completeness, and safety/performance reporting was minimal (Hoogervorst, 2023).
In the UK the National Cardiac Audit Programme (NCAP) reports on 11 cardiovascular sub-specialties (or ‘domains’) which includes a number of registries as outlined in Table 2 with details from the 2025 annual reports:
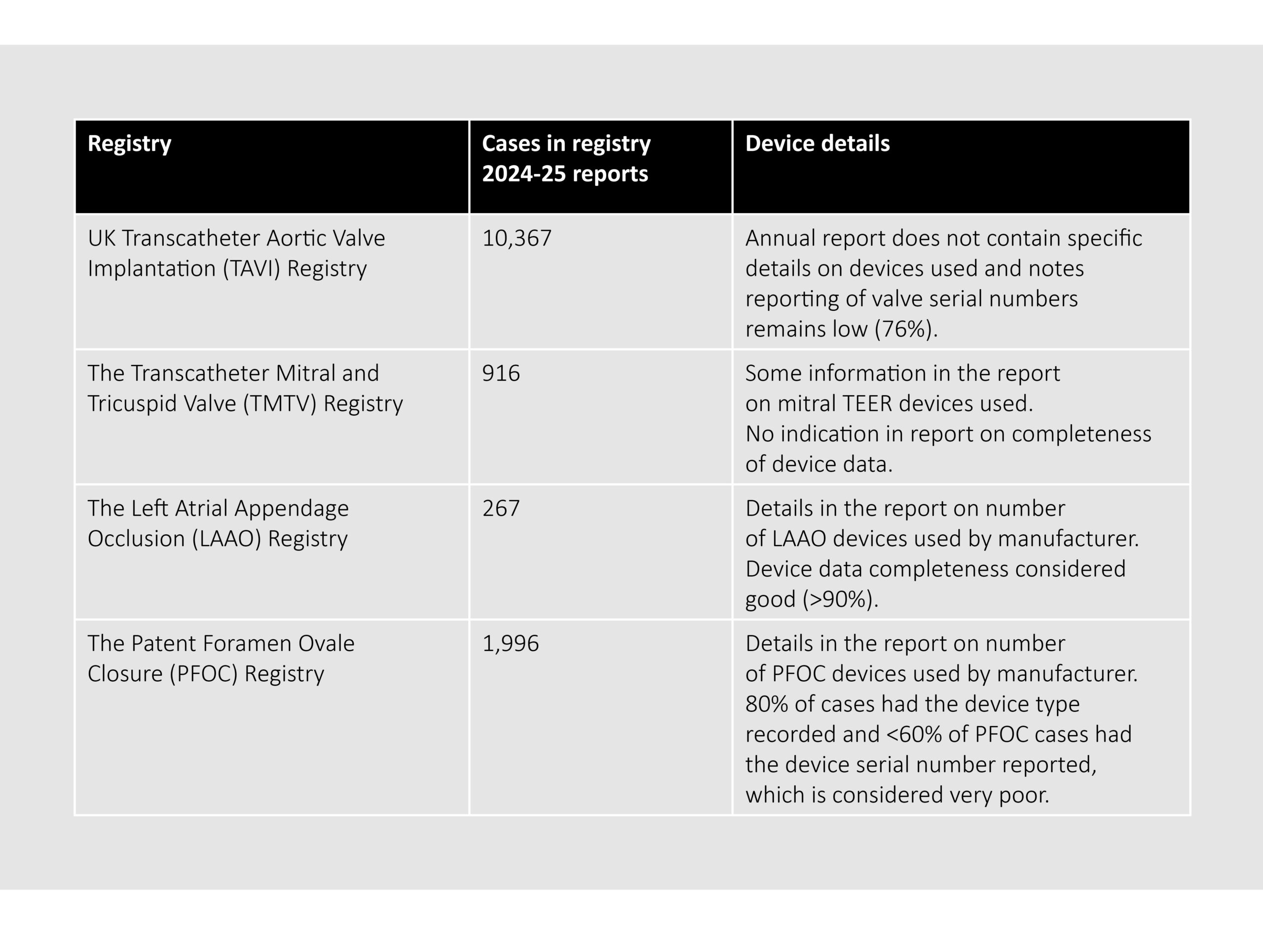
Table 2 – Summary of UK Cardiovascular registry 2025 reports
The NCAP registry reports include information on complication rates, procedures details and length of hospital stay. Due to variability in data completeness and limited device‑specific information, registry data alone may be insufficient to demonstrate that safety and performance requirements are met across all device variants and indications throughout the device lifetime.
The American College of Cardiology have a Voluntary Hospital Public Reporting system for CathPCI, EP Device Implant and Chest pain – MI registries; there do not appear to be public reports listing devices used, however there are publications in the literature (or example Malenka, 2022).
Dental Implants
Few national dental implant registries exist, but emerging data demonstrate their potential regulatory value (Berge, 2025 and Naemi, 2021). In the Norwegian pilot registry, 1,326 implants were registered with a one‑year failure rate of 2.9%. This registry was discontinued in 2006 (as was the Finnish registry in 2016). In Sweden, work towards a national dental implant quality registry started in 2005 and this work is ongoing.
Spine Implants and Spine Surgery Devices
Nine currently active national spine registries collect PROMs (ODI, EQ‑5D, pain scales), complications, reoperations, and device‑specific data (Quigley, 2023 and Pascucci, 2023). Coverage is as high as 98–100% in some Scandinavian registries. Spinal registries include the British Spinal Registry, the Norwegian registry for spine surgery (NORspine), Eurospine (Spine Tango), the Canadian Spine Outcomes and Research Network (CSORN), SweSpine, DaneSpine and the American Spine Registry (AmSR). Table 3 summarises data from spinal registry public reports.
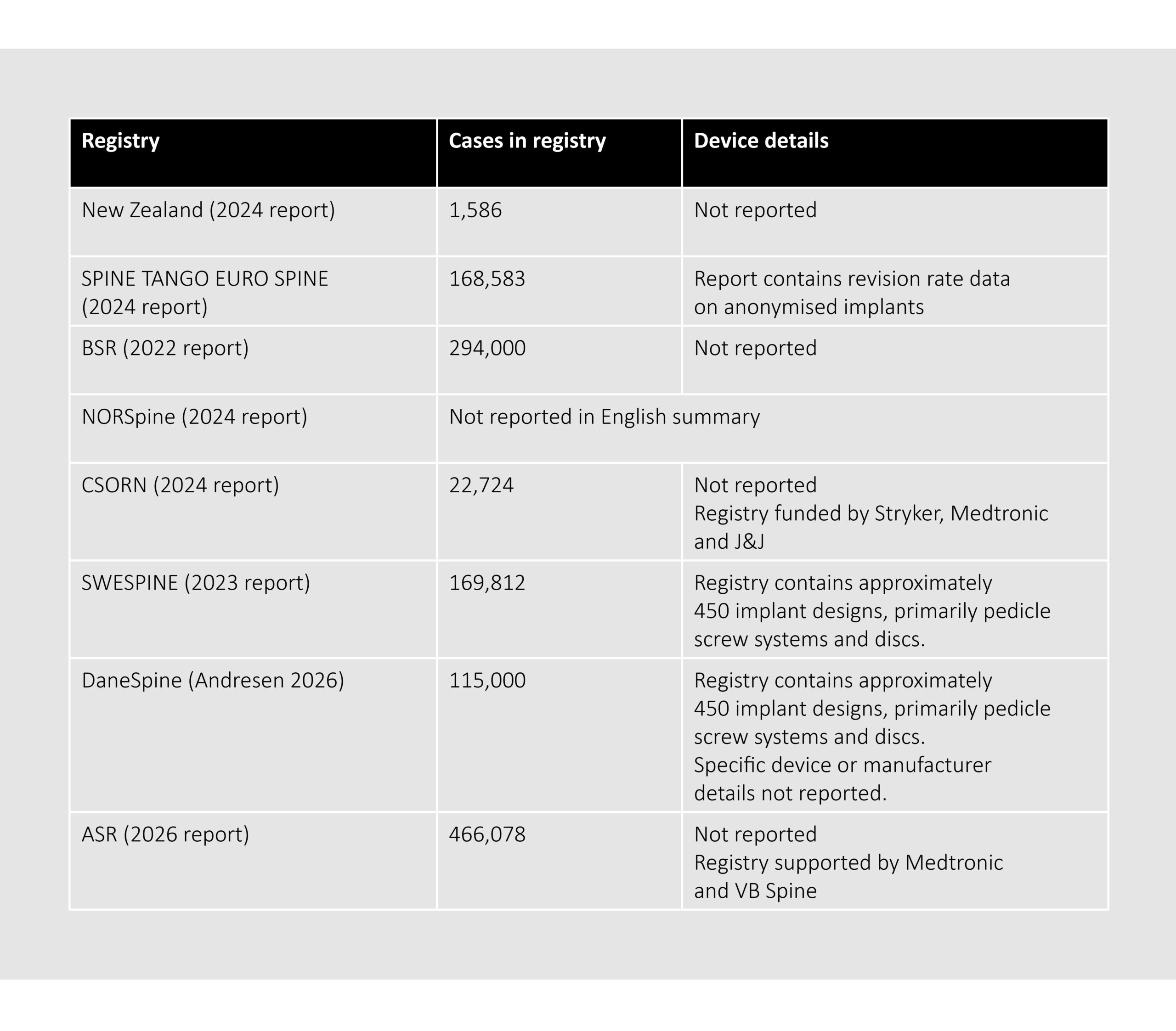
Table 3 – Summary of spinal registry public reports
Public report safety and performance outcomes data is currently variable and inconsistent in these spinal registries; the registries include information such as revision rates and PROMs. Similar to the registries listed in Table 2, it may not be possible to currently use this data alone to meet the relevant clinical GSPRs due to incomplete device, procedure and outcome details. There is also an ongoing project to establish an International Spine Registry.
Other Registries
Other registries have been established, however they do not all produce regular public reports, however publications can be found in the literature on data from these registries (for example Weishorn, 2025). Some of these registries have been recently established (for example in the UK as part of the 2022 Outcomes and Registries Programme as a result of Recommendation 7 of First do no harm, IMMDS Review, 2020, chaired by Baroness Cumberlege) and may issue public reports in the future. These include:
- The German Cartilage Registry
- The UK Knee Osteotomy Registry (UKKOR)
- Breast and Cosmetic Implant Registry (UK)
- National Registry for Hearing Implants (UK)
- Pelvic Organ Prolapse and Stress Urinary Incontinence Registry (UK)
Endnote
Across healthcare areas, including orthopaedics, cardiovascular medicine, and spinal surgery, numerous clinical registries collect large volumes of valuable real‑world data on the performance and safety of medical devices. While these registries provide critical insights, the completeness and quality of the data they capture can vary significantly. Differences in how information is collected, defined, and reported across national and regional registries often create challenges when attempting to compare outcomes for similar procedures or device categories.
Joint arthroplasty registries represent the most mature data sources, providing the most complete and consistent datasets available. These data may be leveraged, for example, to support devices already on the market (for example in certificate renewal submissions or PSURs) in demonstrating continued compliance with the General Safety and Performance Requirements (GSPRs), or to support a new device where an acceptable justification of equivalence can be established, alongside a commitment to robust post‑market clinical follow‑up (PMCF).
In recent years, additional registries have begun to emerge in areas such as cosmetic implants, pelvic meshes, and other high‑visibility device sectors. As these newer registries mature, they have the potential to become powerful contributors to evidence generation throughout the product lifecycle.
Taken together, the growing number of medical device registries and the increasing level of the data they hold, positions them to potentially play a central role in future regulatory strategies. By supporting the ongoing collection of real‑world evidence and strengthening long‑term safety monitoring, registries can help manufacturers meet clinical data requirements and ultimately improve patient outcomes.
References
- Andreas K. Andresen, Karen Højmark, Mikkel Mylius Rasmussen, Michael Nielsen, Charlotte Sommer Meyer, Kresten Wendell Rickers, Mikkel Ø. Andersen, Spine surgery in Denmark 2009–2024: A 15-year overview from the DaneSpine registry, Brain and Spine, Volume 6, 2026, 106028, ISSN 2772-5294, https://doi.org/10.1016/j.bas.2026.106028.
- American Joint Replacement Registry (AJRR): 2025 Annual Report. Rosemont, IL: American Academy of Orthopaedic Surgeons (AAOS), 2025.
- Australian Orthopaedic Association National Joint Replacement Registry Annual Report 2025
- American Spine Registry 2026 Annual Report
- Berge, T. L. L., Lygre, G. B., Kubon, B., & Lie, S. A. (2025). Key factors for a national dental implant registry. Journal of Dentistry, 154, 105603. https://doi.org/10.1016/j.jdent.2025.105603
- Canadian Joint Replacement Registry (CJRR) 2024–2025 annual report
- Hoogervorst, L. A., Geurkink, T. H., Lübbeke, A., Buccheri, S., Schoones, J. W., Torre, M., Laricchiuta, P., Piscoi, P., Pedersen, A. B., Gale, C. P., Smith, J. A., Maggioni, A. P., James, S., Fraser, A. G., Nelissen, R. G. H. H., & Marang-van de Mheen, P. J. (2023). Quality and Utility of European Cardiovascular and Orthopaedic Registries for the Regulatory Evaluation of Medical Device Safety and Performance Across the Implant Lifecycle: A Systematic Review. International journal of health policy and management, 12, 7648. https://doi.org/10.34172/ijhpm.2023.7648
- Left Atrial Appendage Occlusion (LAAO) 2025 2nd Edition – Annual Report
- Lübbeke, A., Hoogervorst, L. A., Marang-van de Mheen, P. J., Prentice, H. A., Rolfson, O., Nelissen, R. G. H. H., Steinbrück, A., McGauran, G., Barea, C., Erikson, K., Pedersen, A. B., Porter, M., & ISAR group (2025). Arthroplasty registries at a glance: an initiative of the International Society of Arthroplasty Registries (ISAR) to facilitate access, understanding, and reporting of registry data from an international perspective. Acta orthopaedica, 96, 116–126. https://doi.org/10.2340/17453674.2024.42706
- David J. Malenka, Deepak L. Bhatt, Steven M. Bradley, David M. Shahian, Jasmine Draoui, Claire A. Segawa, Christina Koutras, Jinnette D. Abbott, James C. Blankenship, Robert Vincent, John Windle, Thomas T. Tsai, Jeptha Curtis, Matthew Roe, Frederick A. Masoudi, The National Cardiovascular Data Registry Data Quality Program 2020: JACC State-of-the-Art Review, Journal of the American College of Cardiology, Volume 79, Issue 17, 2022, Pages 1704-1712, ISSN 0735-1097, https://doi.org/10.1016/j.jacc.2022.02.034.
- National Joint Registry (NJR) – 22nd Annual Report 2025
- Naemi, R., Barikani, H. R., & Shahmoradi, L. (2021). Dental implant quality registries and databases: A systematic review. Journal of education and health promotion, 10, 214. https://doi.org/10.4103/jehp.jehp_1302_20
- Pascucci, S., Langella, F., Franzò, M., Tesse, M. G., Ciminello, E., Biondi, A., Carrani, E., Sampaolo, L., Zanoli, G., Berjano, P., & Torre, M. (2023). National spine surgery registries’ characteristics and aims: globally accepted standards have yet to be met. Results of a scoping review and a complementary survey. Journal of orthopaedics and traumatology : official journal of the Italian Society of Orthopaedics and Traumatology, 24(1), 49. https://doi.org/10.1186/s10195-023-00732-4
- PFOC 2025 2nd Edition Annual Report
- Quigley, M., Apos, E., Truong, T. A., Ahern, S., & Johnson, M. A. (2023). Comorbidity data collection across different spine registries: an evidence map. European spine journal : official publication of the European Spine Society, the European Spinal Deformity Society, and the European Section of the Cervical Spine Research Society, 32(3), 753–777. https://doi.org/10.1007/s00586-023-07529-3
- Spine Tango Annual Report 2024
- TAVI 2025 Annual Report summary
- The British Spine Registry Annual Report 2022
- The New Zealand Joint Registry 26 Year Report 2024
- The Norwegian Registry for Spine Surgery (NORSpine) Annual Report 2024
- The Swedish Arthroplasty Register Annual report 2024
- TMTV 2025 2nd Edition Annual Report summary
- Weishorn, J., Niemeyer, P., Angele, P., Spahn, G., Tischer, T., Renkawitz, T., & Bangert, Y. (2025). Secondary matrix-associated autologous chondrocyte implantation after failed cartilage repair shows superior results when combined with autologous bone grafting: Findings from the German Cartilage Registry (KnorpelRegister DGOU). Knee surgery, sports traumatology, arthroscopy : official journal of the ESSKA, 33(5), 1667–1676. https://doi.org/10.1002/ksa.12467
Disclaimer. The views and opinions expressed in this article are solely those of the author and do not necessarily reflect the official policy or position of Test Labs Limited. The content provided is for informational purposes only and is not intended to constitute legal or professional advice. Test Labs assumes no responsibility for any errors or omissions in the content of this article, nor for any actions taken in reliance thereon.
Get It Done, With Certainty.
Contact us about your testing requirements, we aim to respond the same day.
Get resources & industry updates direct to your inbox
We’ll email you 1-2 times a week at the maximum and never share your information


